
Phosphorus pentabromide PBr5. Chemical reactions YouTube
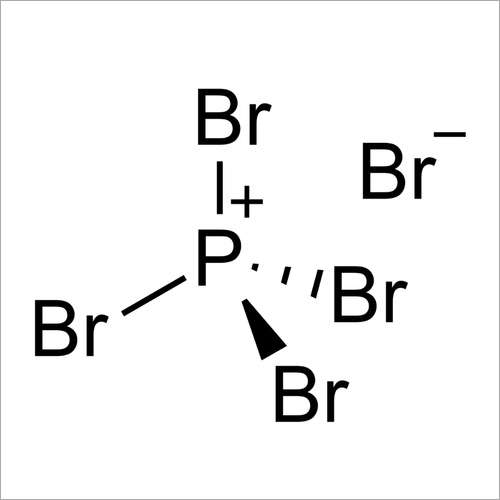
Phosphorus pentabromide is a reactive, yellow solid of formula P Br 5, which has the structure PBr 4 + Br − in the solid state but in the vapor phase is completely dissociated to PBr 3 and Br 2.Rapid cooling of this phase to 15 K leads to formation of the ionic species phosphorus heptabromide ([PBr 4] + [Br 3] −). It can be used in organic chemistry to convert carboxylic acids to acyl.

In this video we are going to learn about the Lewis structure of PBr5
The problem asks to write formula for phosphorus pentabromide. Step 2. 2 of 4. To obtain the formula, we must look at the prefixes on the compound. Phosphorus has no prefix, so we can assume that there is only one atom; while the penta- in pentabromide means that there are five bromine atoms.
Buy Phosphorus Pentabromide at Best Price, Phosphorus Pentabromide
PBr5 is the chemical formula of phosphorus pentabromide, as there are phosphorous and bromine present a 1:5 proportion, as P bears the cationic part so it comes first followed by three bromide ions. PBr5 Structure 3. PBr 5 CAS number

Phosphorus Pentabromide SA.pdf DocDroid
Phosphorus pentabromide is a reactive, yellow solid of formula P Br 5, which has the structure [PBr4]+ Br − (tetrabromophosphonium bromide) in the solid state but in the vapor phase is completely dissociated to PBr3 and Br2.

How to Write the Formula for Phosphorus pentabromide YouTube
It is a chemical formula for Phosphorus Pentabromide.To understand the Lewis. Hey Guys,In this video, we are going to learn about the Lewis structure of PBr5. It is a chemical formula for.
PPT Chemical Bonding PowerPoint Presentation, free download ID6824380
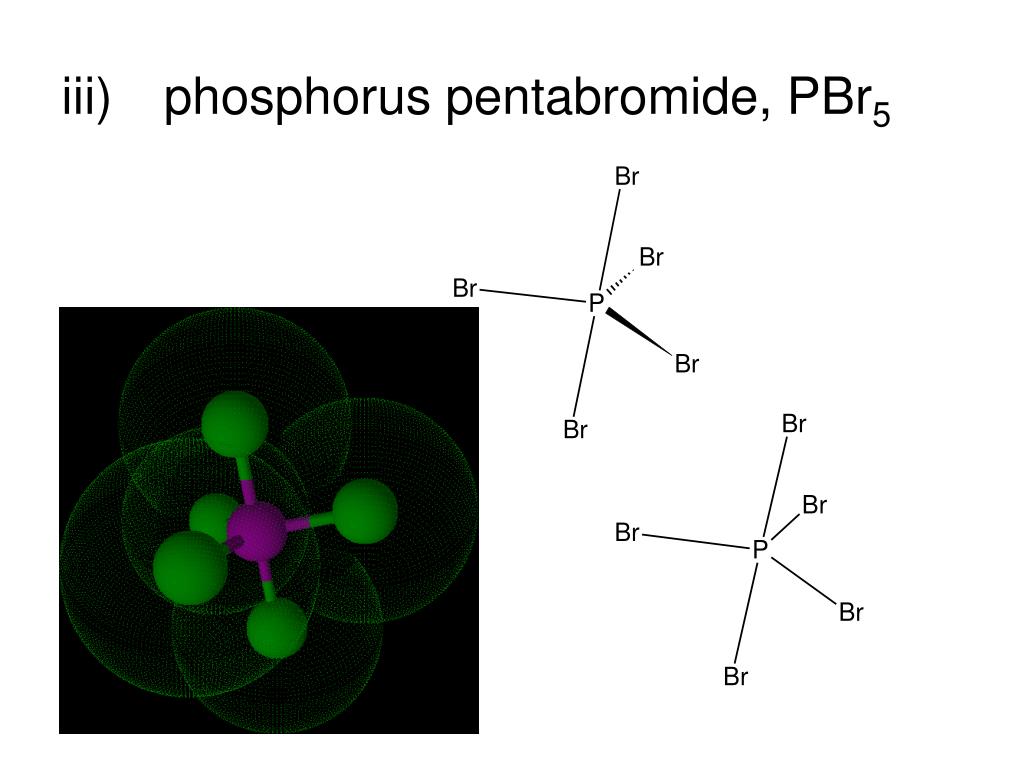
PBr5 or Phosphorous Pentabromide is a compound that consists of 5 molecules of Bromine and 1 molecule of Phosphorus. It appears to be a yellow crystalline solid. The structure of PBr5 in the solid-state is PBr4+ Br− whereas in the vapor phase it dissociates to become PBr3Br2.

ChemistryPhosphorus pentabromide HandWiki
Molecular Formula BrP Average mass 430.494 Da Monoisotopic mass 425.565399 Da ChemSpider ID 56429 More details: Names Properties Searches Spectra Vendors Articles More Names and Synonyms Database ID (s) Validated by Experts, Validated by Users, Non-Validated, Removed by Users 232-186-6 [EINECS] 3D9WIS0BQW 7789-69-7 [RN] MFCD00011437 [MDL number]
Phosphorus pentabromide, 95, Thermo Scientific™
Phosphorus pentabromide is a reactive, unstable, yellow solid chemical compound with the formula PBr5. This compound has the structure PBr 4+ Br − in the solid state, but in the vapor phase is completely dissociated to PBr 3 and Br 2 . Contents [ hide ] 1 Properties 1.1 Chemical 1.2 Physical 2 Availability 3 Preparation 4 Projects 5 Handling

Valency of phosphorus is 3. What is the formula of its oxide, sulphide
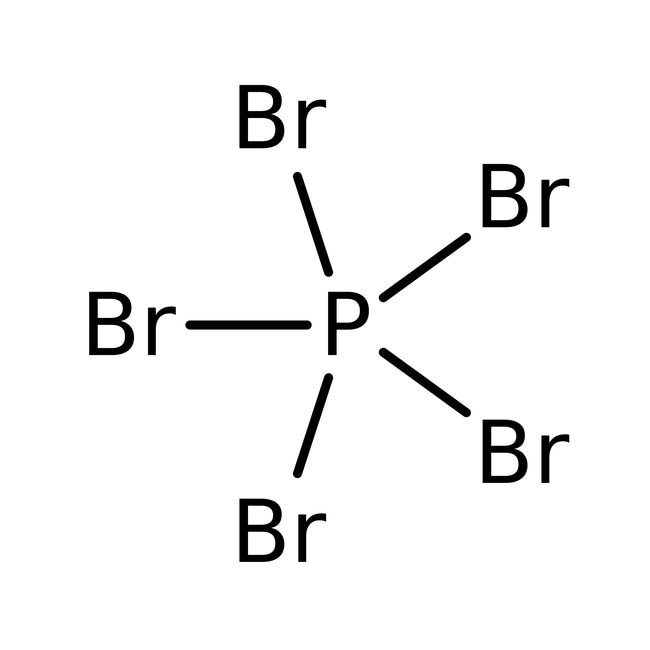
Linear Formula: PBr 5. CAS Number: 7789-69-7. Molecular Weight: 430.49. EC Number: 232-186-6. MDL number: MFCD00011437. PubChem Substance ID: 24853587. NACRES: NA.22.. Phosphorus pentabromide (PBr 5) is a bromination agent generally used to convert alcohols to bromides and in the dibromination of ketones.

Products
0:00 / 1:34 PBr5 Lewis Structure - How to Draw the Lewis Structure for PBr5 Wayne Breslyn 724K subscribers Join Subscribe Subscribed 123 30K views 10 years ago A step-by-step explanation of how.

1 imágenes de Pentabromide Imágenes, fotos y vectores de stock
0:00 / 1:30 How to Write the Formula for Phosphorus pentabromide Wayne Breslyn 718K subscribers 3.4K views 2 years ago In this video we'll write the correct formula for Phosphorus pentabromide.

How to Write the Formula for Tetraphosphorous decoxide (Phosphorous
Phosphorus pentabromide is a reactive, yellow solid of formula PBr5, which has the structure +Br− in the solid state but in the vapor phase is completely dissociated to PBr3 and Br2. Rapid cooling of this phase to 15 K leads to formation of the ionic species phosphorus heptabromide .

Phosphorus Pentabromide Lewis Structure Liqurus
Formula: Br 5 P Molecular weight: 430.494 CAS Registry Number: 7789-69-7 Information on this page: Data at other public NIST sites: X-ray Photoelectron Spectroscopy Database, version 4.1 Options: Switch to calorie-based units Go To: Data from NIST Standard Reference Database 69: NIST Chemistry WebBook

Phosphorus pentachloride Phosphoric acid Chemical compound Chemical
Formula: PBr 5 Hill system formula: Br 5 P 1 CAS registry number: [7789-69-7] Formula weight: 430.494 Class: bromide Colour: orange Appearance: crystalline solid Melting point: >100°C (decomposes) Boiling point: 106°C Density: 3600 kg m -3 The following are some synonyms of phosphorus pentabromide: phosphorus pentabromide
SOLVED(4 points) Write the formula for the following compounds
The Phosphorus pentabromide molecule contains a total of 6 atom (s). There are 1 Phosphorous atom (s) and 5 Bromine atom (s). A chemical formula of Phosphorus pentabromide can therefore be written as: Br5P

H3po4
PBr5 or phosphorus pentabromide is a compound of phosphorus and bromine which is yellow colored (solid) in appearance. It has a got huge application in organic chemistry. So we will study the bonding in PBr5 by understanding the PBr5 lewis structure. Some facts about phosphorus pentabromide This compound has a molar mass of around 430.49 g/mol.